|
|
|
|
|
|
|
|
1. A sealed container contains ice at a temperature well below freezing. Heat is applied at a slow but constant rate. Sketch a graph which shows how the temperature of the container's contents changes over a long period of time as the heat is applied. Label the graph to indicate what is happening in the container.
In this lesson we begin our study of heat in an attempt to see how it fits into the mechanical Newtonian paradigm. Climatic changes have been a major factor in organic evolution and we as a species are extremely sensitive to small temperature changes. Accordingly much of our existence has been concentrated on finding methods to keep warm.
The Greeks learned little of the properties of heat and temperature, and even Aristotle had little to say about it, short of including fire as one of the prime elements. Not much was learned about temperature until the modern ear, beginning with the invention of the thermometer by Galileo. You remember him, don't you?
1.1.1. life is extremely temperature sensitive
1.1.2. location and surface processes keep temperature nearly constant1.1.2.1. just the right distance from the sun
1.1.2.2. daily rotation
1.1.2.3. axial tilt
1.1.2.4. circulation of atmosphere and oceans
1.1.2.5. heat capacity of oceans
1.2.1. small changes have large effects
1.2.2. modern man (homo sapiens sapiens) appears 40,000 years ago during the ice age
1.2.3. technological and cultural adaptation to cold and climate change is a human trait
1.3.1.1. clothing
1.3.1.2. fires
1.3.1.3. heat resistant dwellings
1.4.1. In Aristotle's world heat and cold were basic qualities of matter, and fire was one of four earthly elements
1.5.1. air thermometer also recorded change in atmospheric pressure
1.5.2. showed temperature change but could not measure temperature
1.5.3. not clear who actually invented it, but Galileo wrote about his
Temperature is one of those things that we know so well, yet find it difficult to define. For one thing our perception of temperature is relative. A room at seventy degrees will seem warm to the snowboarder but cold to the tropical surfer. Not only that, but our perception changes with mood and level of activity. A room may suddenly feel warm or cold even though its temperature remains constant.
In terms of our perception, about the best we can do is to say that temperature is a degree of hotness or coldness. Although two people might not agree on whether a particular temperature is hot or cold, they will always agree on which of is two objects is colder or hotter.
2.1.1. hot or cold water
2.1.2. cold tile vs. warm rug
2.2.1. a numerical answer to the question, "How hot is it?"
2.2.2. boiling water is warmer than ice and fire is hotter yet
2.2.3. not very satisfactory definitions
2.2.4. quantitative definition begins with measurement
The measurement of temperature accomplished by using a thermometer (thermo-heat, meter-measure)
Two properties of heat are used in measuring temperature. First is that heat flows from hot to cold objects until they reach the same temperature. This assures that the thermometer will become the same temperature as the thing it is measuring. Second is that there is some measurable thermal property of matter that changes in a regular way when temperature changes.
There are several types of thermometers, the most common being the liquid in glass type.
3.1.1. a thermometer is an instrument which uses some thermal property
3.1.1.1. designed so that we can measure its temperature by observing that property
3.2.1. two objects will eventually reach a common "degree of warmth"
3.2.1.1. the thermometer measures the equilibrium temperature of other bodies with which it is kept in contact
3.2.2. thermal properties of matter change in a regular way with temperature
3.2.2.1. size
3.2.2.2. pressure
3.2.2.3. luminosity
3.2.2.4. color of radiation
3.2.2.5. electrical resistance3.2.3. proportional and easily observed changes are easiest to use

3.3.1. liquid in glass
3.3.2. constant volume
3.3.3. electrical
3.3.4. bimetal
3.3.5. luminosity
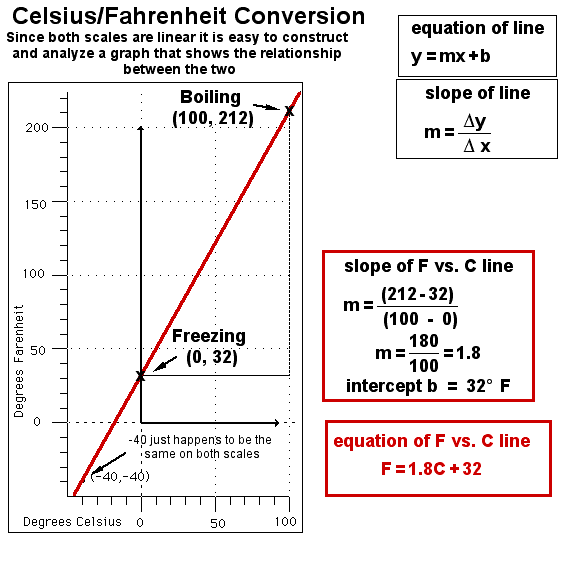
All temperature scales are based on two "fixed points." The scales are arbitrary, just as other units of measurement such as inches. Three scales are in common usage. In physical science we generally use the Celsius system in temperature measurements and the Kelvin scale in temperature calculations. The reason for this will be apparent in later programs. In the Unites States we use the Fahrenheit scale although we are the only country of any size in the world who does. The scales are easily converted back and forth. At the end of this section we will use the properties of linear relationships to show how those conversions can be done.
4.1.1. by Olan Roemer who also made the first accurate light speed measurement in 1675
4.1.2. gave the idea to Fahrenheit who misunderstood the details but got the concept
4.2.1. in the same way that two points determine a line in geometry
4.2.2. primary thermometer will give consistent readings when marked at ice point and steam point

4.2.3. secondary thermometers can be calibrated by immersing in fluid along with primary thermometer
4.3.1. ice/ammonium chloride mixture equals zero degrees
4.3.2. body temperature equals 100 degrees4.3.2.1. Fahrenheit either had a slight fever or inaccurate data
4.3.3. ice point at 32 degrees
4.3.4. steam point at 212 degrees

4.4.1. marked 100 "degrees" between steam point and ice point
4.4.2. steam point equals 100 degrees
4.4.3. ice point equals 0 degrees
4.5.1. same size degree as Celsius scale
4.5.2. ice point 273 K, steam point 373 K
4.5.3. based on gas laws, verified otherwise
4.5.4. to be discussed in detail later
Distinguishing between temperature and heat is one of the most important contributions of physical science. The two concepts are related but distinct. It is difficult to characterize them at this time, but we will do so in more detail in program 26 after we have become better acquainted with atoms and molecules.
At this point let us just say that adding or removing heat from a substance causes a corresponding rise or fall of temperature. We can add that temperature change is proportional to mass for a given substance: twice as much matter requires twice as much heat for a corresponding temperature rise.
5.2.1. when no change of state is involved
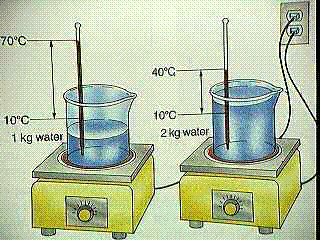
5.3.1. two identical stovetop elements at same temperature will heat a small pan of water faster than large pan
5.3.2. a large stovetop element will heat a given amount of water faster than a small one at the same temperature
5.3.3. 5 kg of water has 5 times as much heat as 1 kg of water at the same temperature
5.3.4. a tub of water can melt more ice than a glass of water at the same temperature
Although temperature change is proportional to mass for a given amount of heat, different substances require different amounts of heat for the same temperature change. This is the first time we have encountered this in our journey. You might be saying, "Wait, didn't Galileo prove that the same laws apply to everything?" The answer is yes. Different substances have the same kind of relationships, but different substances can have different constants of proportion. That is the case with different specific heats.
Specific heat is defined as "thermal capacity per unit mass". It is an intrinsic property of a substance, and is different for each substance. It is based upon the heat unit known as a calorie. The calorie (not a Calorie: the upper case means 1000 calories) is standardized and defined as "the amount of heat required to raise the temperature of one gram of water by one degree Celsius).
Specific heat reflects the chemical and physical differences between different substances at the atomic and molecular level. For that reason we will postpone the explanation for this until program 26.
6.1.1. amount of heat to change temperature of 1 kg of a substance by 1 Celsius
6.1.2. changes in temperature involve heat transfer from hot to cold
6.1.3. establishes quantitative relationship between heat and temperature
6.2.1. equal masses of water and alcohol do not reach the same temperature in equal time

6.2.2. hot iron put into cold water will change temperature more than equal amount of water
6.3.1. defines calorie as heat unit
6.3.2. water has highest specific heat of any substance (except liquid ammonia)6.3.2.1. useful as coolant, moderates climate as oceans store heat
Changes of state (also know as changes of phase) are changes between the various states of matter. The common states are solid, liquid, and gas. Substances change state in response to changes in temperature. The precise conditions of temperature and pressure for these changes are different for different substances.
Changes in state are always accompanied by heat which is either absorbed or given off in the changes. Transitions between the phases can proceed in either direction and may go directly between solid and gaseous states.
The heat given off or absorbed by a substance during a phase change is called latent heat. The latent heat of water is exceptionally large in the change from solid to gas, about 540 calories per gram. (see 6.3 above for definition of calorie). From liquid to solid is also quite high, about 80 calories per gram.

7.1.1. involve absorption or emission of heat
7.1.2. also called phase changes
7.1.3. occur at constant temperature for a given pressure
7.1.4. PT diagrams show ranges of stability of a particular phase, carbon dioxide for example

7.2.1. amount of heat to change 1 kg of substance to another phase
7.2.2. heat of fusion and heat of vaporization

7.2.2.1. a property of a given substance
7.2.2.2. different for different substances7.2.3. reversible physical change
7.2.4. heat absorbed or emitted7.2.4.1. solid --> liquid --> gas: absorbs
7.2.4.2. gas --> liquid --> solid: emits7.2.5. temperature vs. time/heat graph

7.2.5.1. note large latent heats (plateaus) compared to specific heats (slope)
Heat is constantly being transferred from one object to another as heating and cooling take place in the world around us. There are three methods of heat transfer, called "sensible heat" (as opposed to latent heat (see 7.2 above). The effectiveness of each depends on the difference in temperature, the substances, and the situations involved.
These three sensible processes, known as conduction, convection, and radiation operate together, each playing a role proportional to its effectiveness under a given situation. In turn, sensible heat operates along with latent heat to distribute the sun's heat throughout the atmosphere as they do in heating a pan of water on the stove.
8.2.1. dynamic equilibrium
8.2.2. effectiveness depends on substances and situation
8.3.1. convection
8.3.1.1. movement of fluid from one place to another
8.3.1.1.1. heating of water on stove
8.3.1.1.2. wind chill8.3.1.1.2.1. convection increases latent evaporation loss
8.3.1.1.3. winds and deep ocean currents
8.3.1.1.4. large scale geological processes (plate tectonics)8.3.1.2. cannot happen in solids
8.3.1.2.1. slow movement of "solid" earth over long time
8.3.1.3. driven by density differences
8.3.1.3.1.caused by thermal expansion
8.3.1.3.1.1. density = mass/volume
8.3.1.3.2. only in gravitational field
8.3.1.3.2.1. much interest in space stations due to "weightless" environment
8.3.2. conduction
8.3.2.1. conductivity is an intrinsic property of substances
8.3.2.1.1. heat conductors are generally good electrical conductors as well
8.3.2.2. most effective in metals which are good conductors
8.3.2.3. poor in gases which are good insulators or poor conductors8.3.3. radiation
8.3.3.1. any object above absolute zero radiates heat
8.3.3.2. form of electromagnetic (EM) radiation
8.3.3.3. type of radiation depends on temperature
8.3.3.4. wavelength inversely proportional to Kelvin temperature
8.3.3.5. intensity of radiation is proportional to fourth power of Kelvin temperature
8.3.3.6. no material required to transmit8.3.4. insulation is designed to minimize heat flow
8.3.4.1. best insulation addresses all three types
8.3.4.2. thermos bottle is designed to reduce heat loss from all processes

8.3.4.3. styrofoam limits loss by conduction
8.4.1. evaporation/condensation
8.4.2. freezing/thawing
As warm-blooded mammals it is important for us to keep our body temperatures constant within a narrow range of temperatures. An elevated temperature of even two degrees Fahrenheit is enough to send us to the doctor with a "fever". The process of maintaining constant body temperature is known as "homeostasis". Our bodies maintain this temperature by maintaining a balance between heat production and heat loss.
Heat is produced by combustion of food in body tissue. The faster the metabolic rate the more heat is generated. The use of muscles generates heat by speeding up metabolism as does the secretion of hormones by various organs of the body. Heat is also transferred to the body by a fire, the sun, or contact with warm air or water.
Heat loss is controlled in different ways by different animals. Most animals have little tolerance for changes and are dependent on the environment for constancy.
We shiver, move to warmer or cooler quarters, put on or remove clothes, take a hot bath. or a cold dip, sweat, etc.
Dogs pant, cat lick, pigs and hippos bathe.
In all cases animals use various combinations of the sensible heat transfer types to increase or decrease heat input or output from the body. The understanding of heat gained from physical science have helped to understand and monitor heat regulation in animals, ourselves included.
9.1.1. energy comes from chemical energy stored in food
9.1.2. rate of energy usage regulated by biochemical processes
9.2.1. most organisms depend on environmental factors\
9.2.1.1. have very low tolerances
9.2.1.2. a few degrees change may be fatal9.2.2. aquatic life relies on high specific heat of water
9.2.3. balance between metabolic heat and dissipation9.2.3.1. metabolic heat produced through release of chemical energy
9.2.3.2. heat dissipated by various transfer methods9.2.4. insects, reptiles, amphibians
9.2.4.1. cold blooded
9.2.4.2. low metabolic rate
9.2.4.3. supplemented heating and cooling
9.2.4.4. require less food per unit body mass9.2.5. mammals, birds
9.2.5.1. constant body temperature requires homeostasis
9.2.5.1.1. homeostasis: self regulation of constancy
9.2.5.2. regulated by body thermostat, metabolism
9.2.5.3. shivering, changes in blood flow, evaporation, convection, radiation
In this lesson we have explored the nature of temperature and the distinction between temperature and heat. Heat is that which flow from hot to cold and temperature is a measure o f hotness or coldness, both of which are largely subjective. Although we may not agree on whether a particular object is hot or cold, we will all agree on which of two objects feels warmer.
We saw how temperature is measured , how temperature scales were established, and the relationship between various temperature scales.
We defined specific heat and latent heat and learned that different substances have differing responses to the addition of heat, emphasizing that the relationship between heat and temperature involves the concept of the properties of a particular substance.
After a brief review of both sensible and latent heat transfer we looked at the relationship between heat and life.
In the next lesson we will explore the concept of heat as a form of energy and will return to a more satisfying definition and a finer distinction in the program about kinetic theory.